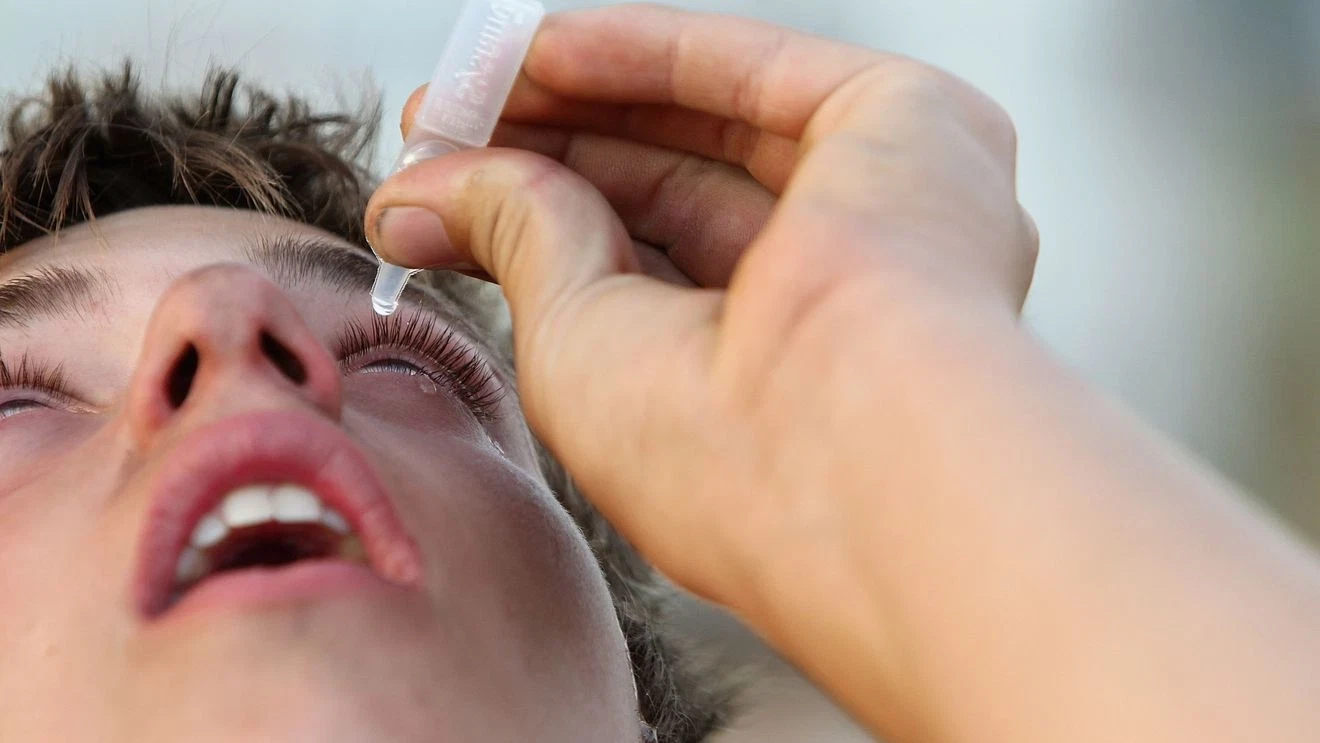
(WKRC) – More than 3.1 million bottles of eye drops sold nationwide are being pulled from shelves after federal regulators flagged concerns about sterility.
K.C. Pharmaceuticals Inc., based in Pomona, California, is voluntarily recalling multiple eye drop products because of a “lack of assurance of sterility,” according to the Food and Drug Administration. The recall affects products distributed across the United States, including through retailers and distributors such as Kroger, Walgreens, H-E-B and Military Exchanges, the FDA said.
The FDA has designated the action as a Class II recall, meaning use of the products could cause temporary or medically reversible health problems.
The recall was initiated March 3 and classified March 31. The FDA lists the recall as ongoing and said a press release was not issued for this recall.
BarkBox - Monthly Dog Subscription Box
Get your puppy the best toys and treats delivered monthly! BarkBox offers premium dog toys, treats, and chews that are perfect for growing puppies. Each box is curated by experts and includes 2 toys, 2 bags of treats, and a chew.
Amazon Puppy Essentials
Everything you need for your new puppy in one place. From crates and beds to food bowls and training pads, Amazon has all the essentials at great prices with fast shipping.
The FDA’s enforcement report includes additional product details such as recall numbers, lot codes, expiration dates, and UPC codes tied to specific store brands and distributors.